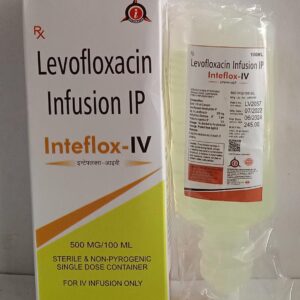
Description
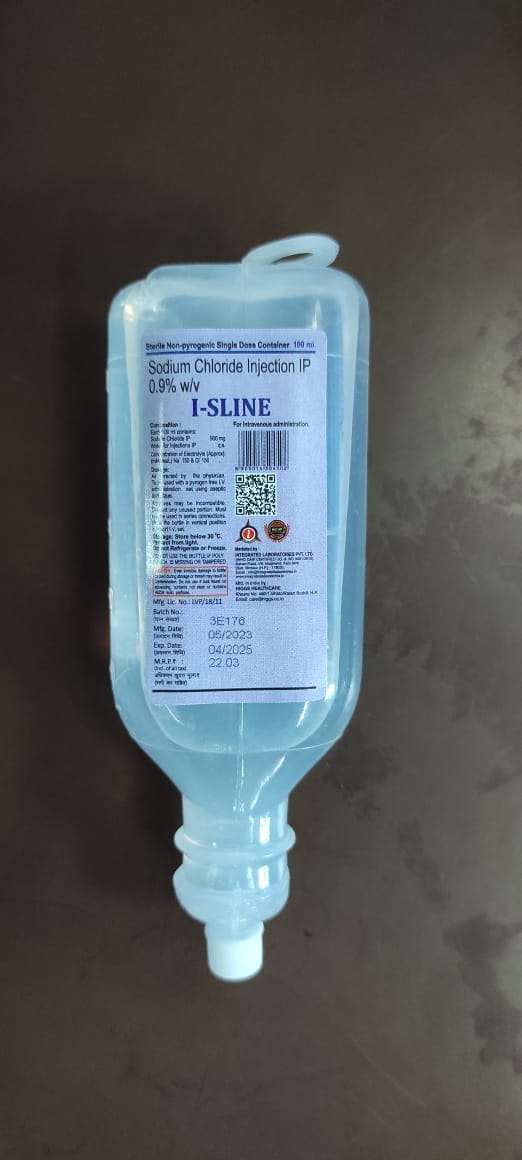
Sodium Chloride 0.9% (I-Sline)
If you are using this product at home, learn all preparation and usage instructions from your healthcare professional. If either is present, do not use the liquid.. Learn how to store and discard medical supplies safel
Click on the “Enquire” button to submit your requirement for Pcd pharma franchise or pharma third party manufacturing of this product.
Side Effects
serious side effects, including swelling hands/ankles/feet, muscle cramps, unusual weakness, headache, nausea, extreme drowsiness, mental/mood changes (such as confusion), and seizures. Redness, pain, or swelling at the injection site may occur. Tell your doctor or pharmacist promptly if any of these effects persist or worsen. Remember that this medication has been prescribed because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.Overdose
If someone has overdosed and has severe symptoms such as passing out or trouble breathing
Missed Dose
It is important to get each dose of this medication as scheduled. If you miss a dose or if your dose is interrupted or stopped, ask your doctor right away for a new dosing schedule.Storage
Store at room temperature away from light and moisture. Do not store it in the bathroom. Keep all medications away from children and pets.
Do not flush medications down the toilet or pour them into a drain unless instructed. Properly discard this product when it is expired or no longer needed. Consult your pharmacist or local waste disposal company.
Also For Higher Antibiotics (Beta-lactam):- Welcureremedies
Explore more from this category:- Multimineral Tablet (Intevit-F) Ondansetron Injection (Insetron) Granisetron 1mg (Integron)
Terlipressin Injection (Interli-10)
Piracetam Injection(I-Ctam)
Tramadol hydrochloride 50 mg/ml (Integrol)
Nandrolone Decanoate Injection (Intebol-25)


Reviews
There are no reviews yet.