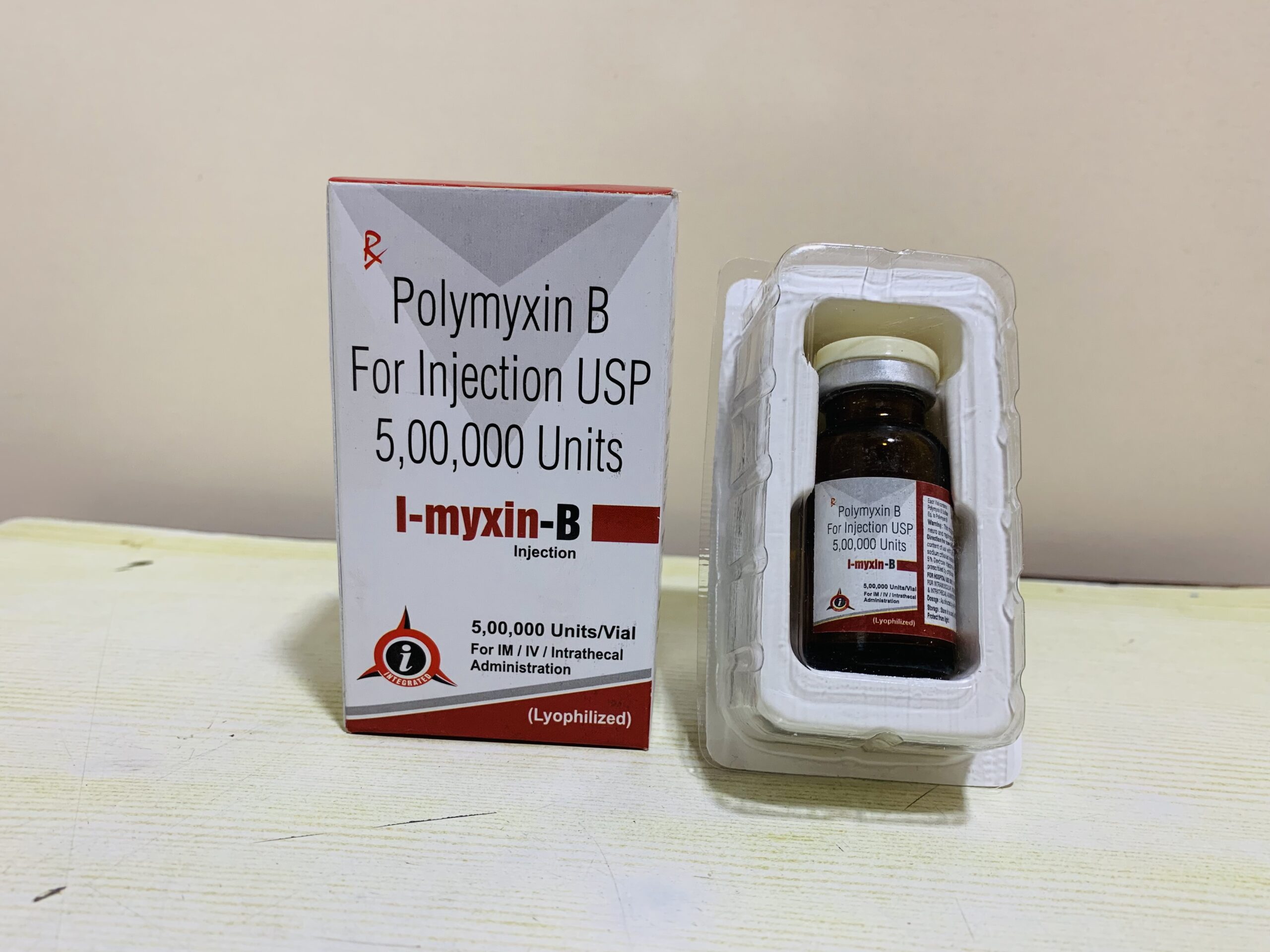
Description
Polymyxin B sulfate Injection (I-myxin-b)
Polymyxin-B Injection is used to treat patients with the following infections when caused by susceptible strains of the designated aerobic gram-negative bacteria:
- Urinary tract infections: Caused by Pseudomonas aeruginosa and Escherichia coli.
- Bloodstream infections: Caused by Pseudomonas aeruginosa, Enterobacter (formerly known as Aerobacter) aerogenes, and Klebsiella pneumonia.
- Meningeal infections: Caused by Pseudomonas aeruginosa.
- Corneal and subconjunctival eye infections: Caused by Pseudomonas aeruginosa.
-
DOSAGE
The recommended dose of Polymyxin B Injection may be administered intravenously, intramuscularly, or intrathecally only to hospitalized patients under constant supervision by a healthcare professional. The polymyxin b dosage should not exceed 2.5 mg/kg/day or 200 mg/day.
SIDE EFFECTSThe most commonly reported side effects of polymyxin B include neurotoxicity, pain at the injection site, electrolyte imbalance, and urticaria.
WARNINGS AND PRECAUTIONS
- Therapy with polymyxin B USP 500000 IU shouldn’t be used in individuals with a prior history of hypersensitivity reactions to it.
- The polymyxin B sulfate Injection, if prescribed for the administration of intravenous, intramuscular, or intrathecal, should be used only in hospitalized patients under precise monitoring for kidney function and neurological signs/symptoms.
- To reduce the development of drug-resistant bacteria and maintain the effectiveness of Inj polymyxin B dose and other antibacterial drugs, it should be used only to treat/prevent infections that are proven to be caused by susceptible bacteria.
- The Inj polymyxin B dose should not be used in patients with myasthenia gravis.
- Polymyxin-B Injection is nephrotoxic; therefore, the renal function should be evaluated before and regularly while on treatment.
- Polymyxin-B sulfate injection should be used with extreme caution in patients with porphyria.
- Polymyxin B uses are restricted during pregnancy unless the expected benefit to the mother outweighs any potential risk to the fetus.
- HOW SUPPLIED/STORAGE AND HANDLING: Care is exercised in the handling of Polymyxin B. It should not be open or broken. Polymyxin B Injection should be stored at temperatures 20°C to 25°C (68°F to 77°F)
- DRUG INTERACTION
- The concurrent use of other nephrotoxic/neurotoxic drugs, particularly bacitracin, streptomycin, kanamycin, tobramycin, cephaloridine, amikacin, cephalothin, polymyxin E (colistin), neomycin, vancomycin, and gentamicin, should be avoided.
- Because of the effect of polymyxin antibiotic drugs on the release of acetylcholine, depolarizing muscle relaxant succinylcholine and other neurotoxic drugs must not be used concurrently with polymyxin sulfate.
- The concurrent use of polymyxin b sulfate with potent diuretics such as ethacrynic acid or furosemide should be avoided since diuretics can enhance polymyxin B sulfate toxicity by altering the antibiotic concentration in serum and tissues.
![Azithromycin Injection [Introcin-500]](https://integratedlaboratories.in/wp-content/uploads/2022/04/Azithromycin-Injection-Introcin-500-300x300.jpg)
Reviews
There are no reviews yet.